Neuralink Gets a Look Inside Your Head
Neuralink wants to bust some heads (in the name of science).
Sign up to uncover the latest in emerging technology.
Neuralink wants to get a better look inside your head. Yes, I’m being literal.
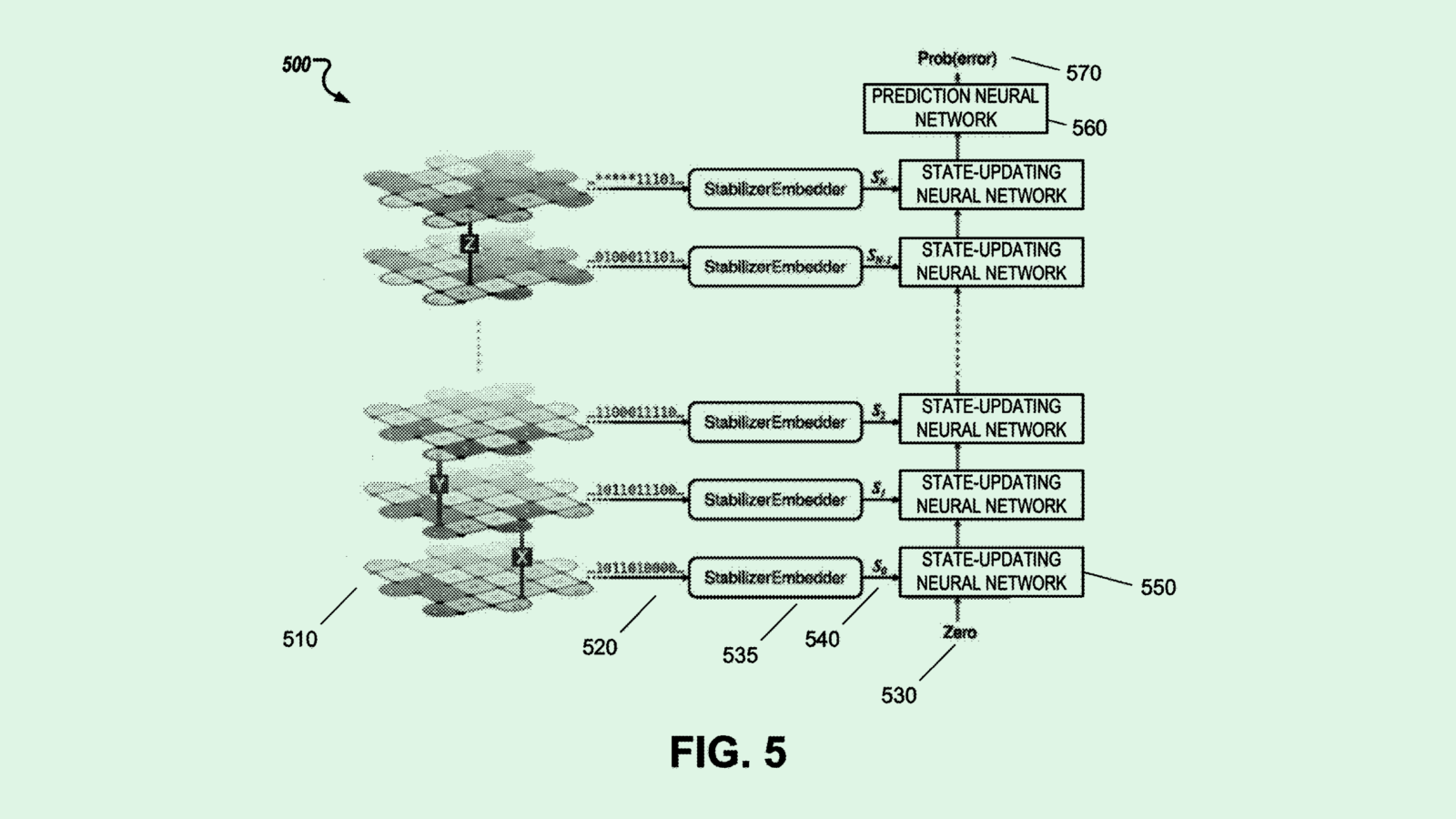
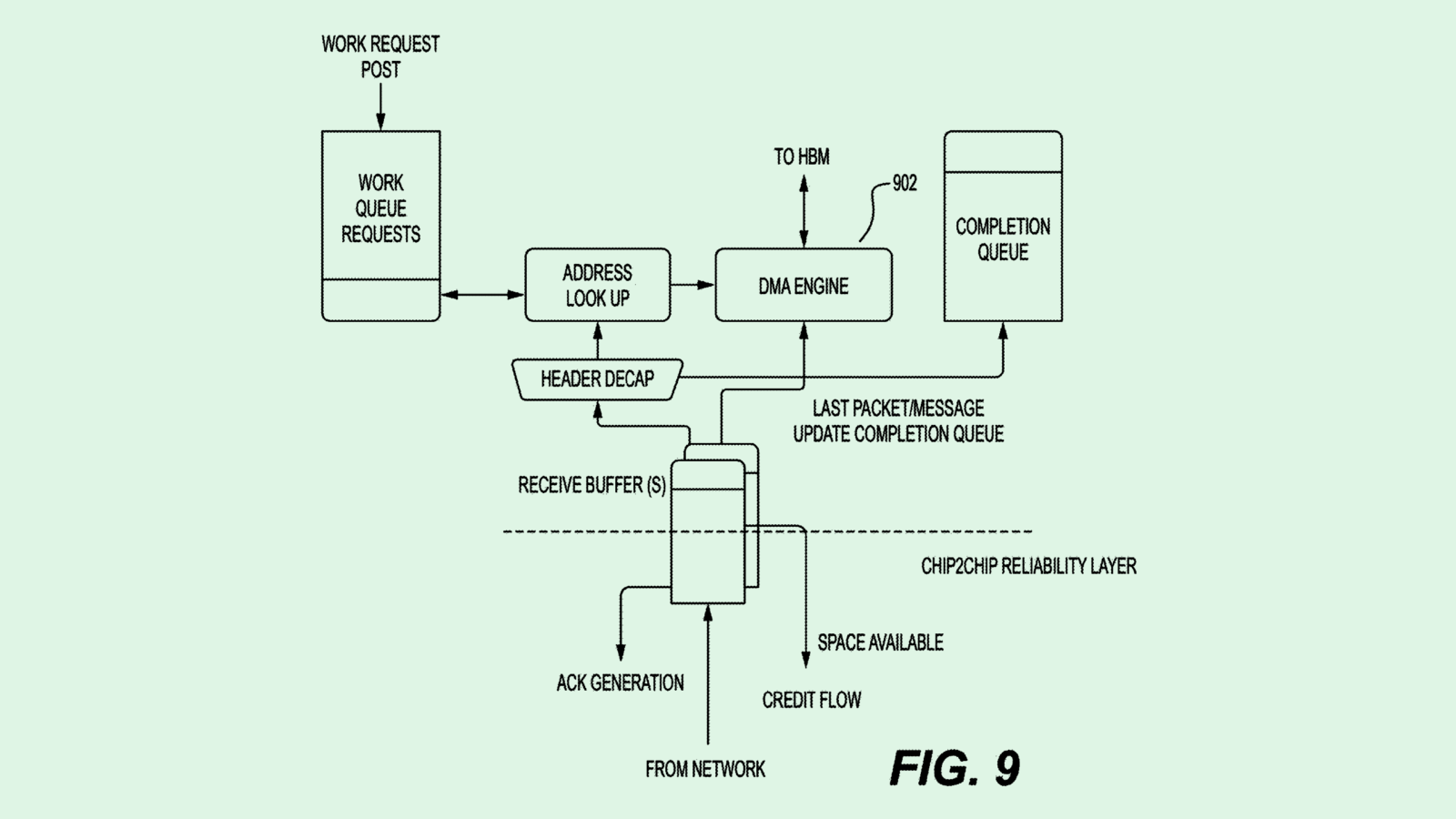
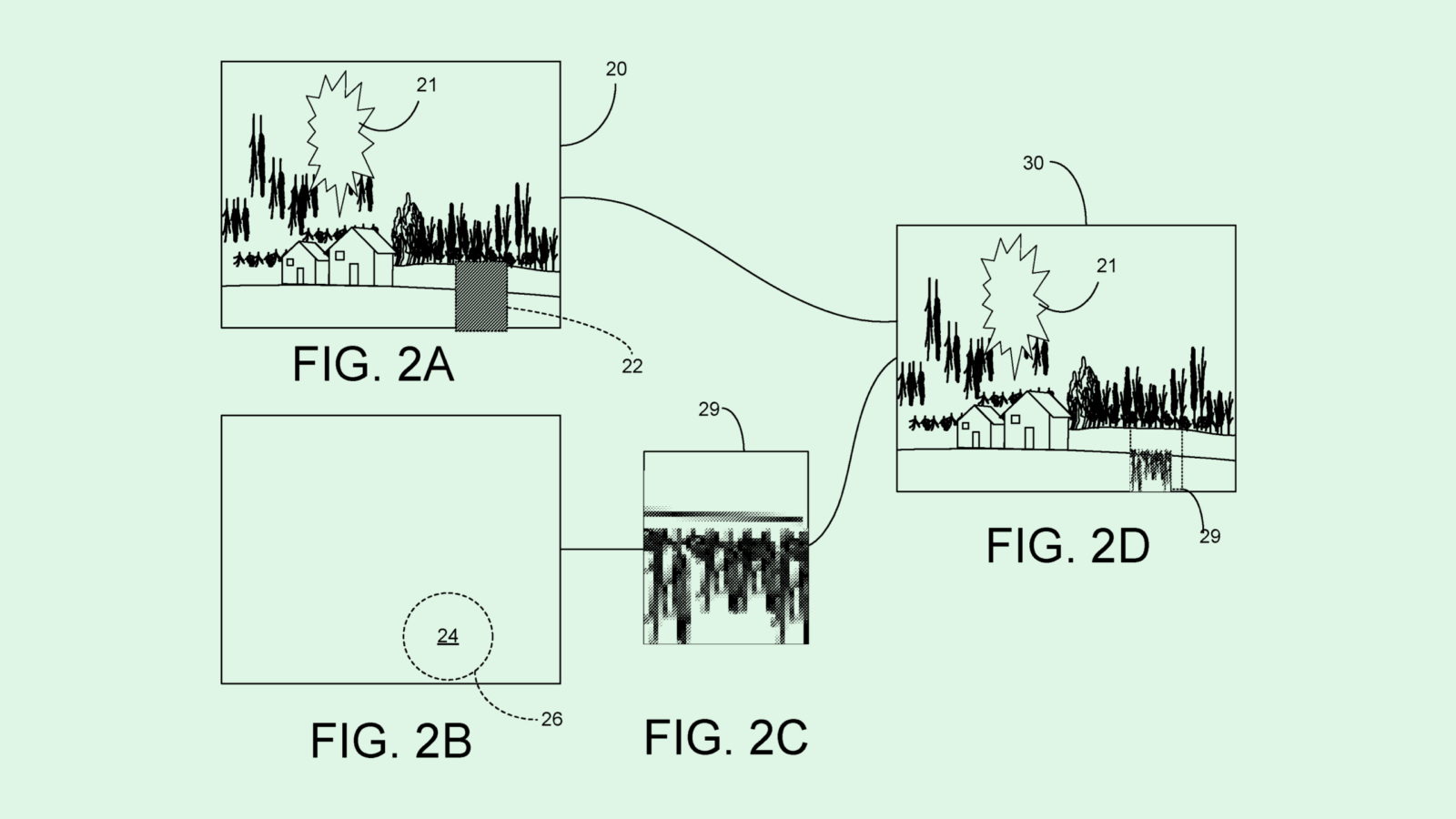
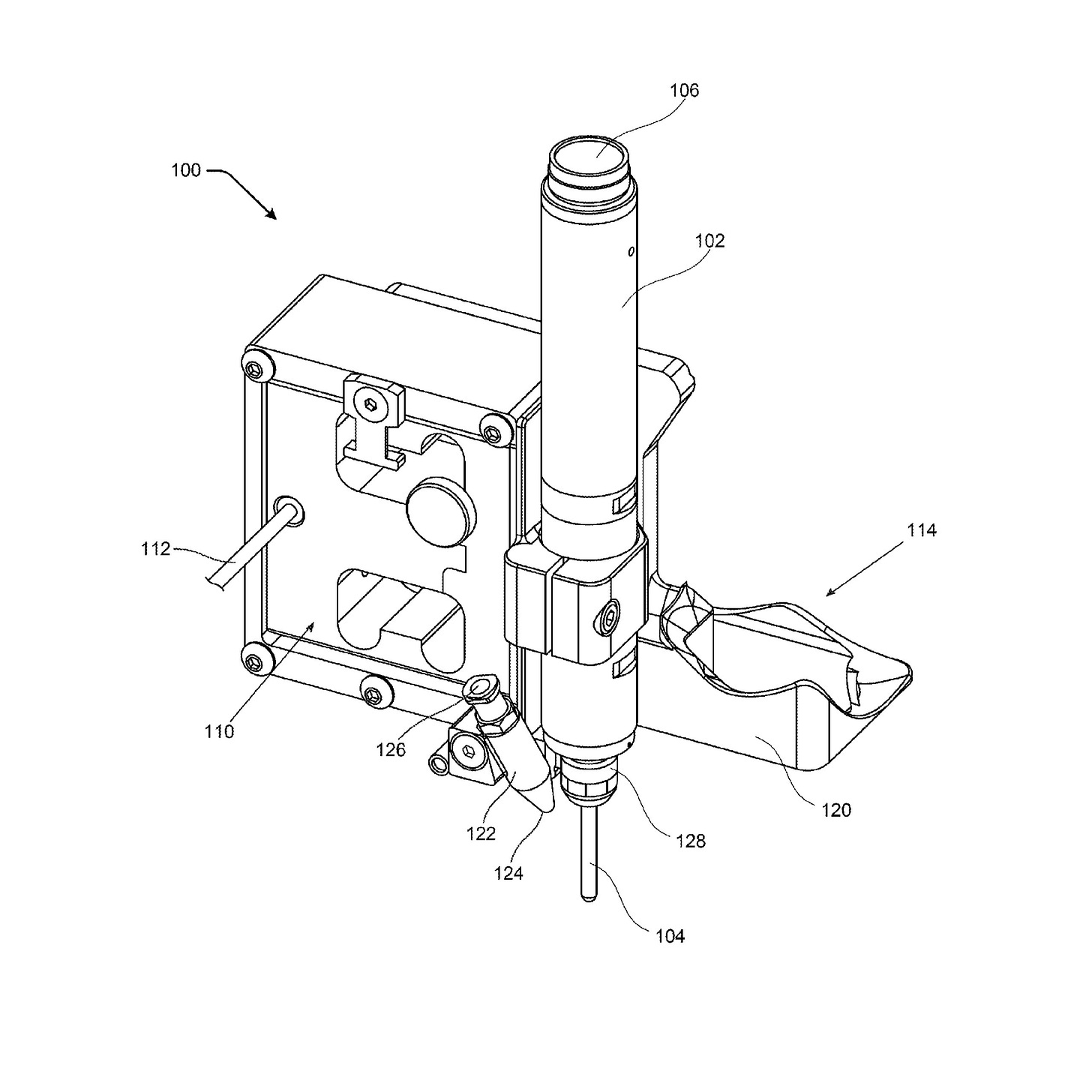
The Elon Musk-owned brain chip company is seeking to patent a method for an “automatic craniotomy” milling system – basically, a machine that precisely performs the surgery of opening the skull. Rather than a surgeon performing this surgery, this machine is operated by a controller that sets the “feed rate,” or how fast the tool is going.
This machine relies on data from two types of sensors: an impedance measurement system (which checks the patient’s response to an electrical current) and an axial force sensor (which senses how much force the machine is using to cut through certain parts of the cranium). Coupled together, these measurements determine the feed rate in response to these measurements.
Neuralink said that traditional craniotomy is often “a time-consuming and high-risk procedure, in part due to the reliance on manual cutting or drilling procedures to create the surgical perforation of the cranium.”
“Neurological implant devices are typically not size adjustable and must securely fit within the craniotomy location to prevent the intracranial structures remaining exposed after implantation,” Neuralink noted. “As such, there is little tolerance for human-caused inaccuracies in the craniotomy procedure.”
Neuralink’s patent offers a lot of technical advantages, said Dr. Georgios Matis, PhD, neurosurgery consultant and head of the Pain/Spasticity department at University Cologne Hospital. Not relying on a doctor’s technical skill in an extremely delicate process improves accuracy and alleviates the risk of human error present in almost any surgery. And a machine which, in real-time, automatically takes measurements into account that would typically be done by hand could allow for a much faster surgery, and could lessen the risk of brain bleeds, seizures, swelling or strokes, he said.
“It seems to be an innovative approach that could contribute to better outcomes,” Matis said in an email. “I am sure many colleagues will be willing to test this technology. However, as with all new patents, it should be widely clinically evaluated.”
Neuralink is inching closer and closer to human trials of its brain-computer interface devices. The company’s request to start clinical trials was first rejected by the FDA in 2022, Reuters reported in March, with the agency highlighting safety concerns related to things like the device’s lithium battery, whether its wires could migrate to other parts of the brain, and the safety of getting the device removed.
However, last week, the company announced that it’s been approved by the agency to conduct its first clinical trial on humans, which Neuralink called an “important first step that one day will allow our technology to help many people.” The company reportedly has investors excited, too: According to Reuters, it reached a valuation of $5 billion.
But the company has a long road ahead of it, Matis said. Neuralink faces a laundry list of barriers that stand in the way of making its chips a widespread reality, he said, including the cost of development, time constraints and labor, proving the product is safe, and the ethical considerations of a device that has the potential to fundamentally change human to human connection.
“It will probably take years (at best) or decades (most probably) till the existing & future research can be safely & with credibility turned into an implantable medical device with no or extremely limited technical problems/limitations, low energy consumption & proven clinical outcomes,” Matis said.
Have any comments, tips or suggestions? Drop us a line! Email at admin@patentdrop.xyz or shoot us a DM on Twitter @patentdrop. If you want to get Patent Drop in your inbox, click here to subscribe.